FIT FOR LIFE MAX RASPBERRY KETONES - Trademark Details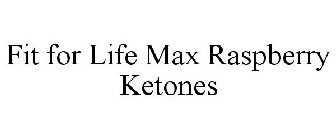
Status: 710 - Cancelled - Section 8
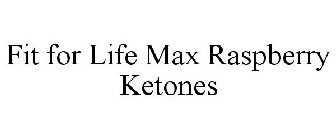
Serial Number
86049320
Registration Number
4702191
Word Mark
FIT FOR LIFE MAX RASPBERRY KETONES
Status
710 - Cancelled - Section 8
Status Date
2021-10-20
Filing Date
2013-08-27
Registration Number
4702191
Registration Date
2015-03-17
Mark Drawing
4000 - Standard character mark
Typeset
Published for Opposition Date
2014-12-30
Law Office Assigned Location Code
L10
Employee Name
HERMAN, RUSS
Statements
Disclaimer with Predetermined Text
"RASPBERRY KETONES"
Goods and Services
Animal feed supplements; Beverages containing chlorophyll for use as a nutritional supplement; Calcium supplements; Dietary and nutritional supplements; Dietary and nutritional supplements containing fish oil, chlorophyll, l-glutamine, inulin, trans-resveratrol, grape seed extract, quercetin, acai berry extract, pomegranate fruit, extract, protease, bromelain, sucrase, raspberry ketone, phosphorus, melatonin, l-theanine, valerian extract, aloe vera gel, ashwagandha, vitamin a, vitamin e, vitamin d, vitamin c, vitamin b1, vitamin b2, thiamin, riboflavin, niacin, vitamin b6, folic acid, biotin, pantothenic acid, calcium, iodine, magnesium, strontium, zinc, selenium, copper, manganese, chromium, potassium, malic acid, d-ribose, co q10, isolated soy lecithin, green tea leaf extract, l-lysine, l-proline, glucosamine, alanine, valine, leucine; Dietary and nutritional supplements for endurance sports; Dietary and nutritional supplements used for weight loss; Dietary beverage supplements for human consumption in liquid and dry mix form for therapeutic purposes; Dietary food supplements; Dietary supplement for eliminating toxins from the intestinal tract; Dietary supplemental drinks in the nature of vitamin and mineral beverages; Dietary supplements; Dietary supplements for animals; Dietary supplements for controlling cholesterol; Dietary supplements for human consumption; Dietary supplements for pets; Dietary supplements for urinary health; Dietary supplements for treating fatigue, sleep problems, humans, animals; Enzyme dietary supplements; Food supplements, namely, anti-oxidants; Herbal supplements; Herbal supplements for sleeping problems; Homeopathic supplements; Lecithin for use as a dietary supplement; Liquid herbal supplements; Liquid nutritional supplement; Liquid protein supplements; Liquid vitamin supplements; Mineral nutritional supplements; Mineral supplements; Mineral, vitamin, or nutritionally enhanced water; Natural supplements for treating candida; Natural supplements for treating erectile dysfunction; Non-medicated additives for animal feed for use as nutritional supplements; Nutraceuticals for the treatment of low energy, treating fatigue, sleep problems; Nutraceuticals for use as a dietary supplement; Nutraceuticals for use as a dietary supplement for digestion, cleansing the liver; Nutritional supplement for eliminating toxins from the body; Nutritional supplement for eliminating toxins from the intestinal tract; Nutritional supplement in the nature of a nutrient-dense, protein-based drink mix; Nutritional supplements; Nutritional supplements consisting primarily of fish oil, chlorophyll, l-glutamine, inulin, trans-resveratrol, grape seed extract, quercetin, acai berry extract, pomegranate fruit, extract, protease, bromelain, sucrase, raspberry ketone, phosphorus, melatonin, l-theanine, valerian extract, aloe vera gel, ashwagandha, vitamin a, vitamin e, vitamin d, vitamin c, vitamin b1, vitamin b2, thiamin, riboflavin, niacin, vitamin b6, folic acid, biotin, pantothenic acid, calcium, iodine, magnesium, strontium, zinc, selenium, copper, manganese, chromium, potassium, malic acid, d-ribose, co q10, isolated soy lecithin, green tea leaf extract, l-lysine, l-proline, glucosamine, alanine, valine, leucine; Nutritional supplements for promoting overall health for humans and animals; Nutritional supplements in the form of liquids, powders, capsules; Nutritional supplements, namely, probiotic compositions; Protein supplements; Vitamin A preparations; Vitamin and mineral supplements; Vitamin B preparations; Vitamin C preparations; Vitamin D preparations; Vitamin enriched water; Vitamin supplements; Vitamins; Vitamins for pets; Weight management supplements; Whey protein supplements; all the forgoing goods containing raspberry ketones
Classification Information
International Class
005 - Pharmaceutical, veterinary and sanitary preparations; dietetic substances adapted for medical use, food for babies; plasters, materials for dressings; material for stopping teeth, dental wax; disinfectants; preparations for destroying vermin; fungicides, herbicides. - Pharmaceutical, veterinary and sanitary preparations; dietetic substances adapted for medical use, food for babies; plasters, materials for dressings; material for stopping teeth, dental wax; disinfectants; preparations for destroying vermin; fungicides, herbicides.
US Class Codes
006, 018, 044, 046, 051, 052
Class Status Code
2 - Sec. 8 - Entire Registration
Class Status Date
2021-10-20
Primary Code
005
First Use Anywhere Date
2013-08-26
First Use In Commerce Date
2013-08-26
Correspondences
Name
ENIVA USA
Address
Please log in with your Justia account to see this address.
Trademark Events
| Event Date | Event Description |
| 2013-08-30 | NEW APPLICATION ENTERED IN TRAM |
| 2013-09-05 | NEW APPLICATION OFFICE SUPPLIED DATA ENTERED IN TRAM |
| 2013-11-23 | ASSIGNED TO EXAMINER |
| 2013-11-27 | NON-FINAL ACTION WRITTEN |
| 2013-11-27 | NON-FINAL ACTION E-MAILED |
| 2013-11-27 | NOTIFICATION OF NON-FINAL ACTION E-MAILED |
| 2014-06-25 | ABANDONMENT - FAILURE TO RESPOND OR LATE RESPONSE |
| 2014-06-25 | ABANDONMENT NOTICE MAILED - FAILURE TO RESPOND |
| 2014-08-21 | TEAS PETITION TO REVIVE RECEIVED |
| 2014-08-21 | PETITION TO REVIVE-GRANTED |
| 2014-08-21 | TEAS RESPONSE TO OFFICE ACTION RECEIVED |
| 2014-08-21 | CORRESPONDENCE RECEIVED IN LAW OFFICE |
| 2014-08-21 | TEAS/EMAIL CORRESPONDENCE ENTERED |
| 2014-08-21 | NOTICE OF REVIVAL - E-MAILED |
| 2014-08-28 | APPROVED FOR PUB - PRINCIPAL REGISTER |
| 2014-09-11 | ASSIGNED TO LIE |
| 2014-09-15 | LAW OFFICE PUBLICATION REVIEW COMPLETED |
| 2014-09-29 | WITHDRAWN FROM PUB OTQR REQUEST |
| 2014-11-20 | PREVIOUS ALLOWANCE COUNT WITHDRAWN |
| 2014-11-20 | EXAMINERS AMENDMENT -WRITTEN |
| 2014-11-20 | EXAMINERS AMENDMENT E-MAILED |
| 2014-11-20 | NOTIFICATION OF EXAMINERS AMENDMENT E-MAILED |
| 2014-11-20 | EXAMINER'S AMENDMENT ENTERED |
| 2014-11-20 | APPROVED FOR PUB - PRINCIPAL REGISTER |
| 2014-11-24 | LAW OFFICE PUBLICATION REVIEW COMPLETED |
| 2014-12-10 | NOTIFICATION OF NOTICE OF PUBLICATION E-MAILED |
| 2014-12-30 | PUBLISHED FOR OPPOSITION |
| 2014-12-30 | OFFICIAL GAZETTE PUBLICATION CONFIRMATION E-MAILED |
| 2015-03-17 | REGISTERED-PRINCIPAL REGISTER |
| 2020-03-17 | COURTESY REMINDER - SEC. 8 (6-YR) E-MAILED |
| 2021-10-20 | CANCELLED SEC. 8 (6-YR) |